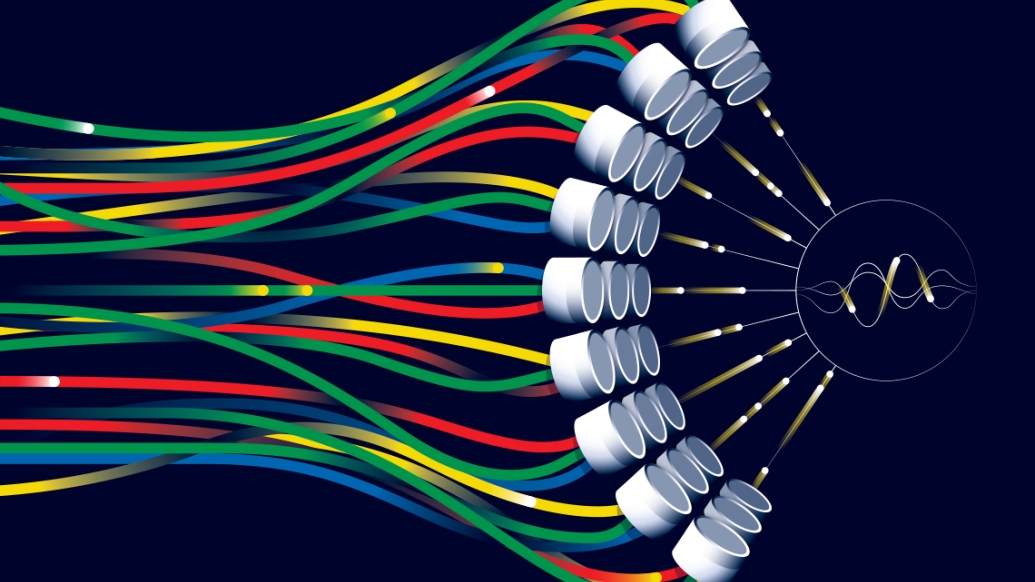
It's a thing of proper science fiction — a technology that can remove tissue without knives or incisions. It has the potential to obliterate tissue without any physical contact, reducing pain and recovery times and improving on the precision of more invasive procedures. And it was invented at the University of Michigan.
The technology, developed by U-M faculty and students over the course of 15 years, is called histotripsy. It's a noninvasive therapy capable of generating ultrasonic pulse sequences powerful enough to break down tissue, but gentle enough to leave the surrounding areas unharmed.
"[Histotripsy means] 'crushing of tissue,'" says J. Brian Fowlkes, M.D., Ph.D., a professor of radiology and of biomedical engineering, associate vice president for research health sciences and a founding member of the histotripsy team.

Noninvasive therapy using ultrasonic pulses is nothing new to medicine, but the process behind histotripsy is. The exact mode of "crushing" is the most essential part. Whereas existing therapies, like high-intensity focused ultrasound, or HIFU, employ thermal energy to ablate tissues, histotripsy harnesses the energy of thousands of microbubbles — called cavitation — to emulsify tissue.
"Cavitation is a phenomenon where ultrasound activates tiny gas bubbles, and those bubbles undergo a collapse. As a consequence of that collapse, there are all sorts of mechanical effects," says Fowlkes.
Initially, cavitation was seen as an adverse side effect of ultrasonic therapy caused by the existence of millions of nanometer-sized gas pockets in the body. In areas of low pressure, these pockets, if hit with enough ultrasonic energy, begin to produce "clouds" of microbubbles that expand and contract in milliseconds.
But cavitation was a nuisance — it was thought to be uncontrollable, and it was something to avoid at all costs.
Fowlkes, whose dissertation examined the biological effects of cavitation, came to the university in 1988 to expand upon his research. It was here that he met and partnered with Charles Cain, Ph.D., the Richard A. Auhll Professor of Engineering, professor of biomedical engineering and of electrical engineering and computer science and an expert in the field of ultrasonic therapy.
Cain had long believed cavitation was much more than a simple side effect, and he was convinced that those thousands of microbubbles could be the genesis of an entirely different mode of treatment. It all hinged on the mechanical activity of the bubbles, which produces kinetic energy.
Purely mechanical energy does not use or produce heat. Instead, it is generated from kinetic or potential energy. In theory, using the mechanical energy of the collapsing bubbles would remedy various limitations existing ultrasonic therapies had. It was easy to describe, but challenging to prove — there existed no means of testing it. So, the idea was initially met with resistance, Fowlkes says. Most colleagues simply couldn't envision any practical method of containing the chaos of bubbles.
Cain, Fowlkes and a handful of students already making advances in the field were poised to prove conventional wisdom wrong.

A Common Purpose
The histotripsy team developed organically. The members were experts in several areas: physics, mathematics, electronics, urology. All of them had a theoretical understanding of the process, but translating that to machinery involved pushing the known limits of ultrasonic technology. They knew that they had to develop something new.
"There was nothing available commercially," explains Tim Hall, Ph.D., associate research scientist of biomedical engineering, a graduate student at the time the work started. "We are doing things outside the normal applications for the electrical parts we use … [so] we have to be creative in finding parts that will fit our needs."
Zhen Xu, Ph.D., associate professor of biomedical engineering and of pediatrics and communicable diseases, was also a graduate student in Cain's lab, having joined the team in 2001. Her work involved finding the exact pulse sequences needed to generate cavitation, using a commercially available ultrasonic transducer with a powerful amplifier. For cavitation, a shorter pulse is needed — shorter, but much more intense — to produce the desired effect. It also requires a significant amount of energy.
Xu labored for months to find the sequence. But, in 2002, she became the very first person to watch controlled cavitation.
"I remember seeing … smoke coming out of the tissue surface while I was experimenting. Within a minute or two, I started seeing perforation. I could actually see a hole," Xu says.
That first visualization confirmed that there was something quite exceptional at work within the bubble clouds, and it led Xu to write paper after paper to establish histotripsy as a process. However, the amplitude of the pulse sequences was still too low to control and optimize the bubble clouds for tissue removal.
Hall's goal was to correct this. Based on Xu's findings in the lab, he developed a custom electrical driving system that could be combined with commercial transducers. The system allowed stock machines to produce the complex pulse sequences needed for histotripsy to be effective.
Hall worked with Will Roberts, M.D., professor of urology and of biomedical engineering, who joined the U-M faculty in 2004 to research ultrasonic therapies. Roberts brought clinical perspective to the team. And, as a urologist, he helped Hall customize the transducer for its first incarnation as a therapy for benign prostatic hyperplasia, or BPH, which affects 80 percent of men over the age of 80.
In 2005, the team combined Hall's custom electronics with a transducer from Imasonic, a company based in France that builds standard lithotripters and ultrasonic transducers. The result was the very first prototype of the histotripsy system.
A Triumph of Theory
All new technology has to undergo preclinical testing to demonstrate efficacy on living tissue. For histotripsy, this meant years of data-gathering in the lab. But it also helped the team better understand how their therapy worked.
The beauty of cavitation, they discovered, is that the rapid motion of the bubbles growing and bursting strains tissue, attacking its structure and causing it to collapse. The targeted tissue then turns into an acellular soup, able to be resorbed and expelled by the body's own processes.
It's a stark contrast to existing ultrasonic therapies. In thermal ablation, the tissue actually "cooks," says Fowlkes, and becomes a stiff, necrotized remnant. The body expels some of it — but not all. The same is true of cryoablation, which freezes tissue. Neither therapy is capable of wholly removing the treated tissue, which can lead to further complications. Histotripsy seems to correct this problem, along with many other drawbacks of ultrasonic therapy.
"Cavitation is a threshold phenomenon," says Roberts. "You have to reach a certain amount of energy before cavitation — before the bubbles cloud — can form."
Beyond that point, the bubbles stop. This means that histotripsy is capable of delivering pinpoint accuracy and produces less collateral damage.
In other therapies, there is also the problem of visualization. High-intensity focused ultrasound and thermal ablation require an MRI scanner to monitor the temperature of the target tissue. A series of calculations are then made to estimate the thermal dose needed, but there is no immediate indication of the treatment.
With histotripsy, however, the bubble cloud is a natural contrast agent, and it's visible on a normal ultrasound imaging system. The emulsification of the tissue is verifiable in real time.
"The bubbles show up on the screen; after you treat, the area turns black, because nothing is there [to scatter the ultrasound for imaging]," says Roberts.
Additionally, because histotripsy uses soundwaves, compensating for the body's natural movements during treatment is a nonissue.
"The speed of sound in tissue is close to one mile-per-second," says Hall. "[T]he sound energy is able to reach the target almost instantly. [Because] each cavitation 'event' happens within a few hundred microseconds, the tissue will only move a very tiny amount … so you can be assured the destruction is in the right location."
From the Bench to the Bedside
The Imasonic transducer used in the preclinical studies was a jumping-off point. The team knew they had to refine and revise the technology before any sort of meaningful clinical tests could be conducted. And, while the Imasonic prototype was a success in preclinical uses, it wasn't without its drawbacks. It was lofty and expensive — too expensive to be practical — and it was only capable of administering treatment for BPH. To be effective, it would need to have flexibility for a variety of locations within the body, as well as for a variety of tissues. It also took months to customize, build and ship. The turnaround time wasn't feasible for the number of experiments the team needed to conduct.
A lot of what we envision histotripsy to be able to do is what we've been able to observe. It's incredible … to see the powerful potential [histotripsy] has. —Christine Gibbons
Cain, Hall and Xu decided it would be more cost-effective to build their own transducers for testing purposes. With the advent of 3-D printing, that became a viable option. "If you can adapt the equipment to be optimal for the application or experiment you want to perform, then you have a much better chance of being successful," Hall says. "You wouldn't try to build a car using only three-quarter inch bolts."
Spurred on by the findings in the preclinical studies, the team has begun to delve into uses for histotripsy in different areas of medicine. And, with customized transducers built by 3-D printing and Hall's customized electronics, they can do this quickly and economically.
Currently, Xu is paving the way for applications in liver cancer, pediatric cardiology and thrombolysis. Cain is targeting histotripsy in the brain. Hall and Roberts are focusing on urological applications beyond BPH, including kidney stones and prostate cancer. And, as more collaborators are brought into the fold, more ideas develop. "We now have … a list of people who are willing to cooperate and help us understand the problems that we could use histotripsy to try to solve," Fowlkes says.
The five inventors of histotripsy — none of whom had experience in the business world — are also helping to bring histotripsy to the clinic. In 2008, they worked closely with the U-M Office of Technology Transfer, which provides entrepreneurial support for faculty-led research groups, to found HistoSonics, Inc. The company is currently conducting human trials for BPH at U-M and in Toledo, Ohio. Since July 2013, they have treated 28 patients in a safety pilot trial with encouraging results.
"A lot of what we envision histotripsy to be able to do is what we've been able to observe," says Christine Gibbons, CEO of HistoSonics. "It's incredible … to see the powerful potential [histotripsy] has."
With HistoSonics making strides in clinical translation and the histotripsy team developing and collaborating on more and more uses, that potential seems limitless. For the five researchers and dozens of students told countless times that cavitation was a nuisance, it's especially liberating.
"We invented histotripsy, and we're still the world leader," says Xu. "We want to revolutionize the field of tissue ablation and surgery."